Imaging Driven Innovation
Learn more about recent advances and software tools to leverage medical imaging data in the product lifecycle.
Are you new to working with medical imaging data as an engineer? First you should find out if you will have access to DICOM or an other 3D or 4D medical imaging data format. If so, the complete range of software solutions are potentially available to you. If not, it’s not the end of the world. You probably have an STL or equivalent file type. You have options, just not as many. The outline below summarizes the possibilities:
- STL files: Meshlab and FEMAP (there’s still a lot you can do!)
- Medical Imaging data and NX CAE
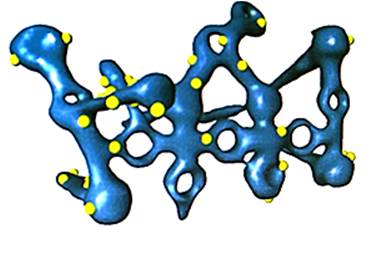
- Automatic segmentation analysis
- Manual segmentation analysis
- Smart segmentation analysis
Contact us to learn more about leveraging medical imaging analysis to improve your product and accelerate it’s development. We can help you with generic patient population data to get you. Use our experience to plan a Smart clinical trial that anticipates your next regulatory hurdle.
Look forward to an upcoming YouTube video series:
- Introduction: Using Medical Imaging Analysis to increase the value of your product
- Open options for Medical Imaging Data (STL => FEMAP)
- Automatic AND Accurate, Siemens PLM i2i in the Orthopedic Industry
- NX Flex Manual Segmentation: Case Study I*
- NX Smart Segmentation: Case study II, Heart Valve. multi-structures for FSI
- Custom solutions, application: Virtual Clinical Trials
- In this segment we explore the design of a ~10 patient study to gather data to be used in the product development process to improve sizing thus avoiding additional clinical trials. Applications of Siemens Healthcare’s Manual and Smart segmentation technology will be used to build models with patient specific imaging data to demonstrate a coupled fluid structure interaction analysis.
* My preferred case study would be a vena cava filter with a monster thrombus trapped and the resulting forces during flow but also tidal breathing loading conditions. even changing orientation and gravity would have an effect on the results. dislodgement and tilting are potential issues with vena cava clot capturing devices and patient specific outcomes.
I have at least 7 other case study ideas, but I think we only need one or two, and it wouldn’t take too much to work those up into good demos.
Well designed clinical trials and thoughtful data collection may be the only difference between having the data you need and regulatory approval.