Peru Workshop on Medical Device Regulation: Policy and Technical Aspects
 We were recently invited to be a keynote speaker for Medical Device Standards: Technical Training on Cardiovascular Materials and Device Aspects at the PERU Workshop on Medical Device Regulation: Policy and Technical Aspects, November 2-4, 2015.
We were recently invited to be a keynote speaker for Medical Device Standards: Technical Training on Cardiovascular Materials and Device Aspects at the PERU Workshop on Medical Device Regulation: Policy and Technical Aspects, November 2-4, 2015.
The event was hosted by ASTM International Standards Alliance with USAID/ANSI. The primary audience was government regulators, national medical device technical committee, medical device users, purchasers from hospitals, companies and testing laboratories. It was a great learning experience and in hope the start of greater fairness and effectiveness of healthcare for everyone.
We presented on three topics:
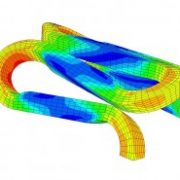
1) Standard Guide for Finite Element Analysis (FEA) of Metallic Vascular Stents Subjected to Uniform Radial Loading ASTM F2514.
2) Standard Test Methods for in vitro Pulsatile Durability Testing of Vascular Stents, plus Shelf-life Test Attributes for Endovascular Devices ASTM F2914-12.
3) Standard Guide for Testing Absorbable Stents, ASTM F3036-13 and Current Issues in Materials, Fatigue and Fracture and Testing of Implantable Devices
Joined by other ASTM colleagues, Kenneth R. St. John, PhD, and his experience with biocompatability and biological test methods and Jon Cammack, PhD, with an overview of TC194 and ISO 10993.
If you would like to see our presentations, please send us a request.