Stent Performance Characterization
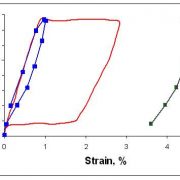
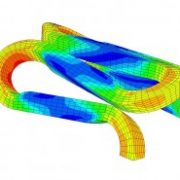
Radial force behavior is one of many engineering design criteria important to the design of implantable medical devices such as stents. Interpretation and extrapolation of bench-top data to predict in-vivo performance necessitates a thorough understanding of influence parameters such as boundary conditions and material uncertainty. An integrated modeling and test validation approach is needed to fully understand interactions of the influence parameters. This understanding helps establish reliable fatigue prediction methodologies that combines the load and displacement type boundary conditions. The approach is more broadly applicable to the unusual and complicated superelastic transformation behavior observed in Nitinol.
You can download our paper presented at the Symposium on Implant Mechanics during the Society for Experimental Mechanics 2007 Spring Conference where we discuss stent radial force performance, FEA, contact issues, experimental and material parameters associated with different radial force test methods and much more: