In vivo Strain Estimates for Medical Implants
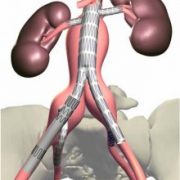
 Defining the in vivo loading conditions for a new device presents great challenges for design validation. Complex implant geometries, multi-axial displacements, device/tissue interactions and time dependent factors can make it difficult or impossible to identify a single loading cycle that is representative of the full range of conditions.
Defining the in vivo loading conditions for a new device presents great challenges for design validation. Complex implant geometries, multi-axial displacements, device/tissue interactions and time dependent factors can make it difficult or impossible to identify a single loading cycle that is representative of the full range of conditions.
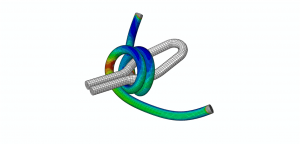
In a paper published in ASTM-STP1559, Fatigue and Fracture of Medical Materials and Devices, we describe a methodology for using synchronous biplane cineflouroscopy imaging data to estimate the in vivo strains for a novel implant design. With tools available at the time, we developed a semi-automated process of converting medical imaging data capable of handling about 10 or so cases.
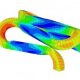
The reconstructions were matched to finite element models of the implant and we studied the fatigue cycle in great detail. We carried out numerous validation checks and uncertainty quantifications and learned an enormous amount about the boundary conditions of a very complex system.
We learned that a small amount of medical imaging data can go a long way. With just five or six cases we established parameters defining the critical fatigue motions for the implant. Today, we can fully automate the data conversion and consider patient studies on the order of 10s and 100’s.
To learn more about the engineering application of medical imaging data, download our In Vivo Strain Estimates for Medical Implants paper